- Internal Audit Reviewer Checklist
- Internal Audit Checklist Examples
- Internal Audit Checklist Template
- Internal Audit Workpaper Review Checklist
- Internal Audit Quality Assurance Review Checklist
- Internal Audit Review Report
Do you want to create a flexible, risk-based audit program — but don’t know where to start? Read our Internal Audit Planning Checklist to learn which audit steps and requirements you should keep in mind, and download our full guide to “Planning an Audit from Scratch” below!
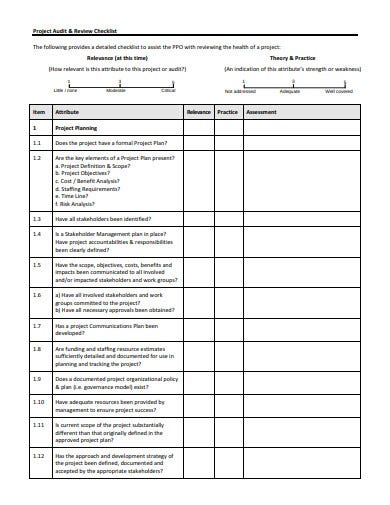
Internal Audit Planning Checklist 1. Initial Audit Planning. All internal audit projects should begin with the team clearly understanding why the project was put on the audit plan. The following questions should be answered and approved before fieldwork begins: Why was the audit project approved to be on the internal audit plan? The internal audit program shall address all elements of the management system, including the testing and/or calibration activities. It is the responsibility of the quality manager to plan and organize audits as required by the schedule and requested by management. Internal Quality Management System Audit Checklist (ISO9001:2015) Q# ISO 9001:2015 Clause Audit Question Audit Evidence 4 Context of the Organization 4.1 Understanding the organization and its context 4.1q1 The organization shall determine external and internal issues that are relevant to its purpose.
What can internal auditors do to prepare a more comprehensive scope for their internal audit projects? And where can internal auditors find the subject matter expertise needed to create an audit program “from scratch”? AuditBoard’s “Planning an Audit from Scratch: A How-To Guide” details how to build an effective internal audit plan from the ground up through best practices, resources, and insights, rather than relying on template audit programs.
One of the guide’s highlights is a comprehensive checklist of audit steps and considerations to keep in mind as you plan any audit project. Use the checklist below to get started planning an audit, and download our full “Planning an Audit from Scratch: A How-To Guide” for tips to help you create a flexible, risk-based audit program.
Internal Audit Planning Checklist
1. Initial Audit Planning
All internal audit projects should begin with the team clearly understanding why the project was put on the audit plan. The following questions should be answered and approved before fieldwork begins:
- Why was the audit project approved to be on the internal audit plan?
- How does the process support the organization in achieving its goals and objectives?
- What enterprise risk(s) does the audit address?
- Was this process audited in the past, and if so, what were the results of the previous audit(s)?
- Have there been significant changes in the process recently or since the previous audit?
2. Risk and Process Subject Matter Expertise
Performing an audit based on internal company information is helpful to assess the operating effectiveness of the process’s controls. However, for internal audit to keep pace with the business’s changing landscape and to ensure key processes and controls are also designed correctly, seeking out external expertise is increasingly becoming a best practice.
At least one of the following should be used to evaluate the design of the process audited:
- Subject Matter Expert (SME) from a Big 4 or other consulting firm
- Recent articles from WSJ.com, HBR.org, or other leading business periodicals
- Relevant blog posts from The Protiviti View, RSM’s Blog, or the IIA’s blogs
Once you have leveraged internal and external resources to identify relevant risks, you will want to build an audit program that tests for these risks.
3. COSO’S 2013 Internal Control – Integrated Framework
While used extensively for Sarbanes-Oxley (SOX) compliance purposes, internal auditors can also leverage COSO’s 2013 Internal Control – Integrated Framework to create a more comprehensive audit program. In addition to identifying and testing control activities, Internal audit should seek to identify and test the other components of a well controlled process.
- Review COSO’s 2013 Internal Control components, principles, and points of focus here.
4. Initial Document Request List
Requesting and obtaining documentation on how the process works is an obvious next step in preparing for an audit. The following requests should be made before the start of audit planning in order to gain an understanding of the process, relevant applications, and key reports:
- All policies, procedure documents, and organization charts
- Key reports used to manage the effectiveness, efficiency, and process success
- Access to key applications used in the process
- Description and listing of master data for the processes being audited, including all data fields and attributes
After gaining an understanding of the process to be audited through the initial document request, you should request access to master data for the processes being audited to analyze for trends and to aid in making detailed sampling selections.
5. Preparing for a Planning Meeting with Business Stakeholders
Before meeting with business stakeholders, internal audit should hold an internal meeting in order to confirm the high-level understanding of the objectives of the process or department and the key steps to the process. The following steps should be performed to prepare for a planning meeting with business stakeholders:
- Outline key process steps by narrative, flowchart, or both, highlighting information inflows, outflows, and internal control components
- Validate draft narratives and flowcharts with subject matter experts (if any)
- Create an initial pre-planning questionnaire to facilitate a pre-planning meeting with key audit customers
Preparing the questionnaire after performing the initial research sets a positive tone for the audit, and illustrates that internal audit is informed and prepared. Once this research is completed, internal audit should meet with their business stakeholders to confirm their understanding of the process.
6. Preparing the Audit Program
Once internal audit has confirmed their understanding of the process and risks within the process, they will be prepared to create an audit program. An audit program should detail the following information:

- Process Objectives
- Process Risks
- Controls Mitigating Process Risks
- Control Attributes, including:
- Is the control preventing or detecting a risk event?
- Control frequency (e.g. daily, weekly, monthly, quarterly, etc.)
- Does the control mitigate a fraud risk?
- Is the control manually performed, performed by an application, or both?
- An initial assessment of the risk event (e.g. high, medium, or low)
- Testing Procedures for Controls to be Tested During the Audit, including:
- Inquiry, or asking how the control is performed
- Observation, or physically seeing the control be performed
- Inspection, or reviewing documentation evidencing the control was performed
- Re-performance, or independently performing the control to validate outcomes
7. Audit Program and Planning Review
Internal Audit Reviewer Checklist
Audit programs, especially those for processes that have never been audited before, should have multiple levels of review and buy-in before being finalized and allowing fieldwork to begin. The following individuals should review and approve the initial audit program and internal audit planning procedures before the start of fieldwork:
- Internal Audit Manager or Senior Manager
- Chief Audit Executive
- Subject Matter Expert
- Management’s Main Point of Contact for the Audit (i.e. Audit Customer)
Internal auditors who can create and document audit programs from scratch — and do not rely on template audit programs — will be more capable and equipped to perform audits over areas not routinely audited. When internal audit can spend more of their time and resources aligned to their organization’s key objectives, internal auditor job satisfaction will increase because they’ll be taking on more interesting projects. The Audit Committee and C-suite may become more engaged with internal audit’s work in strategic areas. Perhaps most importantly, recommendations made by internal audit will have a more dramatic impact to enable positive change in their organizations.
Want to learn more tips to help you create a flexible, risk-based audit program? Download our free 'Planning an Audit from Scratch: A How-To Guide' below.
If your facility is struggling with developing a properly risk based internal audit plan within resource limitations, you are not alone. With the myriad of changes that happen each year in healthcare and the subsequent risks that are presented, it is imperative to develop an internal audit plan that fits the needs of your organization.
An internal audit plan is simply a plan to provide a review of an organization’s processes and procedures over selected areas to ensure financial and operational controls are in place and assure they are mitigating risk for the organization. In healthcare, a common key component of any internal audit plan is compliance-related risks. The Office of Inspector General (OIG) has stated there are basic elements when constructing the compliance portion of your internal audit plan, one of which is conducting internal monitoring and auditing focused on billing and coding issues through performance of periodic reviews. This component of the compliance plan can provide documented evidence to the OIG in their assessment of whether reasonable efforts were taken to avoid fraud and abuse.
Three Main Types of Risk
- Fraud
- Falsified Documentation
- Money Laundering
Basic Steps for Billing and Coding Audits
We have created a checklist below to ensure best practices are met:Establish an Auditor
We recommend the reviews be performed by qualified and credentialed coding and billing staff. If your facility does not have an internal qualified reviewer, an independent consultant should be utilized. If your facility does choose to perform coding and billing reviews internally, we recommend that an independent review be conducted on an annual basis.
The determination of who will perform the audits is an internal organizational decision. Some organizations have the resources to perform quarterly audits as mentioned above, and ensuring the personnel have the proper credentials and have some level of independence is important for an effective audit process in this area. For other organizations, having the proper resources available is not feasible, in which case, an external resource can be engaged. In other cases, a blend of both may be more appropriate. The organization can engage with an audit partner to assist when needed or for specific types of audits.
Determine the Frequency of the Reviews
Best practice states to perform reviews at least on a quarterly basis. If significant issues are noted, this may require more frequent reviews. Once issues are identified, and resolution is agreed to by all parties involved, a plan should be put in place to discuss appropriate actions to resolve errors noted and/or provide necessary education to the appropriate staff. Also, follow up audits should be considered, particularly if a higher level of errors are identified.
Best practice states to perform reviews at least on a quarterly basis. If significant issues are noted, this may require more frequent reviews. Once issues are identified, and resolution is agreed to by all parties involved, a plan should be put in place to discuss appropriate actions to resolve errors noted and/or provide necessary education to the appropriate staff. Also, follow up audits should be considered, particularly if a higher level of errors are identified.
Establish an “Accuracy” Goal
A minimum accuracy threshold needs to be established by the organization. A coding and billing audit can serve as a baseline and identify any areas with a high error rate. Reviews can also identify areas of weakness that can serve as a basis for education and remediation of issues identified.
A minimum accuracy threshold needs to be established by the organization. A coding and billing audit can serve as a baseline and identify any areas with a high error rate. Reviews can also identify areas of weakness that can serve as a basis for education and remediation of issues identified.
An accuracy goal gives the organization some parameters to reach towards. Once audits are completed, these accuracy goals will allow for prioritization for follow-up audits and assist in determining where educational resources should focus. Each area identified in the review should be assigned a risk category based on severity. Here is a suggested guide:
Internal Audit Checklist Examples
| Degree of Risk and Priority of Action | |
|---|---|
| Severe | Notes an issue identified that if not addressed immediately has a high likelihood of impacting the completion of the organization’s strategic or operational objectives. |
| High | A finding with a high probability of material adverse effects that requires immediate action in order to address the concern and reduce risk to the organization. |
| Medium | A finding with a medium probability of material adverse effects that requires management action in order to address the concern and reduce risk to the organization. |
| Low | A finding with a low probability of material adverse effects that requires management to address the concern and reduce risk to the organization. |
| Insignificant | A finding identified by auditors with no risk or impact but typically represents informational or educational purposes. |
Internal Audit Checklist Template
Establish the Audit Size
Sample size is essential in determining accuracy. Smaller sample sizes may not pick up coding and billing error trends. For professional reviews, we typically recommend auditing 10 encounters per provider. This allows for a comprehensive review of a variety of levels. For hospital inpatient/outpatient reviews, the sample size would be dependent on the size of the organization and the volume of patients seen within the various departments.
Sample size is essential in determining accuracy. Smaller sample sizes may not pick up coding and billing error trends. For professional reviews, we typically recommend auditing 10 encounters per provider. This allows for a comprehensive review of a variety of levels. For hospital inpatient/outpatient reviews, the sample size would be dependent on the size of the organization and the volume of patients seen within the various departments.
As noted, quarterly audits of randomly chosen encounters/cases are considered best practice. However, depending on the size of the organization, including all outpatient hospital claims in the population sample will likely be unrealistic. Narrowing the focus to specific areas—the Emergency Department, encounters or a specific type of high volume surgery, for example—may be an option based upon the situation. The focused areas/topics can be planned out for each quarter in advance while allowing flexibility in case an area of concern arises during the audit period.
Selection Process
There are multiple methods for choosing audit areas to review. One common method is to follow the OIG Work Plan. In the past, the OIG came out with a yearly work plan, identifying areas of concern. However, as of June 15, 2017, the OIG has changed their process. There will no longer be an annual workplan, instead the OIG website will be updated monthly to ensure it aligns with evolving issues. New additions will be found under the “Recently Added” tab on the website, while completed Work Plan items will be removed. Monthly additions will ensure that audit cases are timely and relevant. For instance, the OIG website includes a section for Active Work Items. As of March 2019 the list includes:
There are multiple methods for choosing audit areas to review. One common method is to follow the OIG Work Plan. In the past, the OIG came out with a yearly work plan, identifying areas of concern. However, as of June 15, 2017, the OIG has changed their process. There will no longer be an annual workplan, instead the OIG website will be updated monthly to ensure it aligns with evolving issues. New additions will be found under the “Recently Added” tab on the website, while completed Work Plan items will be removed. Monthly additions will ensure that audit cases are timely and relevant. For instance, the OIG website includes a section for Active Work Items. As of March 2019 the list includes:
Internal Audit Workpaper Review Checklist
- Identifying Opioid Misuse
- Billing Off the Shelf Orthotic Devices
- Hospital Billing for Severe Malnutrition
- Payments for Podiatry and Ancillary Services
- Duplicate Payments for Home Health Services
Other methods of choosing cases could include the following:
- Monitor the Center for Medicare and Medicaid Services (CMS) guidelines and transmittals
- Sign up for notifications and guidance from your local Medicare Administrative Contractor (MAC)
- Review denial information which will provide insight into areas where there may be potential compliance risk
- Conduct reviews for new services lines or areas of growth
- Other targeted regulatory audits- UPIC, RAC, etc.
Flexibility should also be allowed for new service lines that are starting within the organization. An evaluation of the coding as well as a review of the claim should be performed to ensure that all elements of the coding and billing are processing correctly.
Internal Audit Quality Assurance Review Checklist
Issue a Report of Findings
Once the review is completed, the appropriate billing and coding staff should have the opportunity to review the detail results and offer any rebuttals and/or explain their rationale for decisions made in the billing process. After all the information has been reviewed and rebuttals taken into consideration, a final summary should be provided to the appropriate responsible individuals within the billing and coding area, along with the internal audit and compliance officer. Problem areas are now identified, an action plan developed, and appropriate education should be provided to coding and billing staff.
Once the review is completed, the appropriate billing and coding staff should have the opportunity to review the detail results and offer any rebuttals and/or explain their rationale for decisions made in the billing process. After all the information has been reviewed and rebuttals taken into consideration, a final summary should be provided to the appropriate responsible individuals within the billing and coding area, along with the internal audit and compliance officer. Problem areas are now identified, an action plan developed, and appropriate education should be provided to coding and billing staff.
Once an initial report of findings has been reviewed and shared with the applicable departments, the internal audit team, working with the compliance department, will want to examine the findings and identify any trends or errors that require urgent attention. For example, if a department is billing for infusion services without documenting start and stop times, a focused review may need to take place sooner, rather than later. There should be some flexibility built into the internal audit plan to allow for these types of focused reviews. However, action plan and remediation should also be established to ensure appropriate and timely resolution of issues based upon degree of risk in the area, and these action plans should be followed up on to ensure risk areas are appropriately resolved.
In our experience, a 95% accuracy rate is required when under a Corporate Integrity Agreement. It’s best practice to apply this same level of accuracy when determining the organization’s acceptable threshold for error. If the findings of the review show less than a 95% accuracy rate, procedures should be in place to address the issues noted. Oftentimes, an action plan outlining the issue and recommendations can serve to engage the key stakeholders, establish deadlines and provide a roadmap with accountability for issue resolution. An example action plan may look like this:
| Issues/Findings | Actions Taken/To be Taken | Responsible Party | Timeline |
|---|---|---|---|
| Improper billing of IV infusion and drug administration charges. Misuse of hierarchy and modifier 59. | Identification of the departments that utilize these charges. Outline of the charge capture process for these areas. Determination of coding involvement and focused education provided for proper coding and modifier assignment. | Revenue Integrity, Chargemaster Coordinator, Coding Manager and specified clinical areas. | Completed 3/11/2019 following identification of these errors. |
| Issue: Improper principal diagnosis code sequencing and code assignment in sepsis-related cases. | Review of the Coding Guidelines and Coding Clinic guidance related to principal diagnosis code assignment, secondary diagnosis code assignment and proper coding for sepsis specifically. | Coding Manager | To be completed by second quarter 2019 |
In the example above, certain findings may encompass solely the coding team while other findings may pertain to a different department. It is not unusual for multiple departments or areas to overlap with respect to billing and coding responsibility for certain pathways. Therefore, it is important to include all the relevant parties in the process early, along with input on finding, resolution and action plan for remediation.
Internal Audit Review Report
In conclusion, we believe a partnership approach between the organization (compliance department) and the internal audit team is essential. Whether one leader is responsible for both areas or separate leaders are working together, communication is key. Internal audits can identify potential risk areas and can help to recommend improvements to mitigate risk. Organizations are encouraged to perform routine internal audits to identify opportunities and provide staff education.
Recorded Webinar
Want to learn more? Check out the recap of our webinar on incorporating coding and billing into your internal audit and compliance plan!
Want to learn more? Check out the recap of our webinar on incorporating coding and billing into your internal audit and compliance plan!
